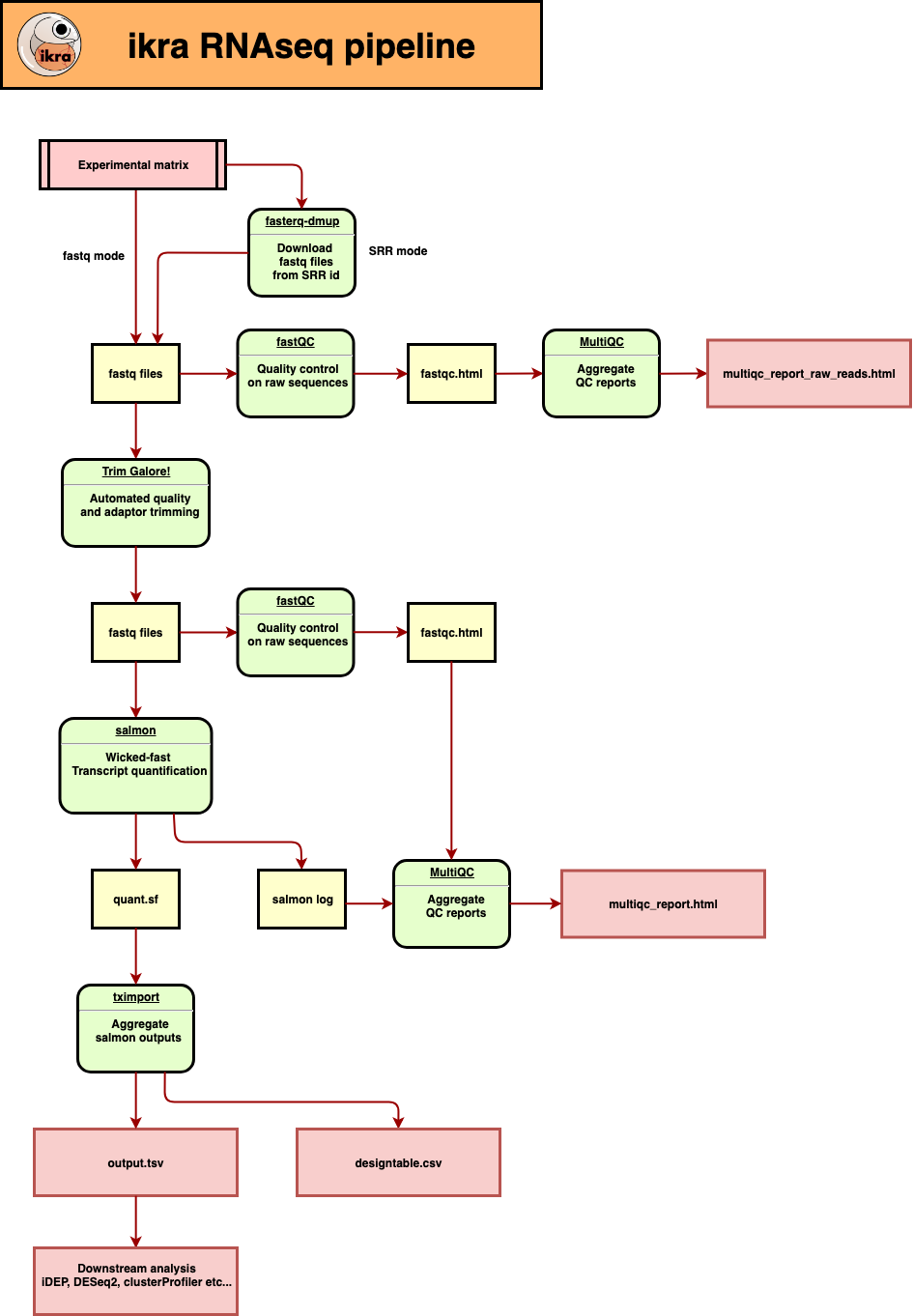
A gene expression table (gene × sample) is automatically created from the experiment matrix. The output can be used as an input of idep. Ikra is an RNAseq pipeline centered on salmon.
Note that sra-tools has to be installed locally. This is up to NCBI's tool upgrade. Please install sra-tools (>=2.10.7).
Usage: ikra.sh experiment_table.csv species \
[--test, --fastq, --help, --without-docker, --udocker --protein-coding] \
[--threads [VALUE]][--output [VALUE]]\
[--suffix_PE_1 [VALUE]][--suffix_PE_2 [VALUE]]
args
1.experiment matrix(csv)
2.reference(human or mouse)
Options:
--test test mode(MAX_SPOT_ID=100000).(dafault : False)
--fastq use fastq files instead of SRRid. The extension must be foo.fastq.gz (default : False)
-u, --udocker
-w, --without-docker
-pc, --protein-coding use protein coding transcripts instead of comprehensive transcripts. (default : True)
-ct, --comprehensive-transcripts use comprehensive transcripts instead of protein coding transcripts. (default : False)
-t, --threads
-o, --output output file. (default : output.tsv)
-l, --log log file. (default : ikra.log)
-a, --align carry out mapping onto a reference genome. hisat2 or star (default : None)
-g, --gencode specify the version of gencode. (defalut : Mouse=26, Human=37)
-s1, --suffix_PE_1 suffix for PE fastq files. (default : _1.fastq.gz)
-s2, --suffix_PE_2 suffix for PE fastq files. (default : _2.fastq.gz)
-h, --help Show usage.
-v, --version Show version.
-r, --remove-intermediates Remove intermediate files
- test option limits the number of reads to 100,000 in each sample.
- udocker mode is for server environments that can only use User privileges. For more information https://github.com/indigo-dc/udocker.
- without-docker mode works with all tools installed. Not recommended.
- protein-coding mode restricts genes to protein coding genes only.
- threads
- output is
output.tsvby default. - align mode generates genome-mapped bam and bigwig files. Note that Salmon works quasi-alignment mode similarly as no-align mode. Generated bw files can be visualized on IGV or other genome browsers. experiment matrix should be separated by commas (csv format).
SRR mode
| name | SRR | Layout | condition1 (optional) | ... |
|---|---|---|---|---|
| Treg_LN_1 | SRR5385247 | SE | Treg | ... |
| Treg_LN_2 | SRR5385248 | SE | Treg | ... |
fastq mode
| name | fastq(PREFIX) | Layout | condition1 (optional) | ... |
|---|---|---|---|---|
| Treg_LN_1 | hoge/SRR5385247 | SE | Treg | ... |
| Treg_LN_2 | hoge/SRR5385248 | SE | Treg | ... |
- Denote names by connecting conditions and replicates with underscores. See idep's Naming convention in detail.
- The first three columns are required.
- If you want to use your own fastq file, add
--fastqoption. Ikra supports only.fq,.fq.gz,.fastqandfastq.gz. - fastq file specifies path excluding
fastq.gzor_1.fastq.gzand_2.fastq.gz. For example,hoge/SRR5385247.fastq.gzis described ashoge/SRR5385247. - If suffix is not
_1.fastq.gzor_2.fastq.gz, add -s1 and -s2 options. - It is impossible for docker to specify a hierarchy above the execution directory, such as
../fq/**.fastq.gz, but it can be avoided by pasting a symbolic link. bonohu blog
- output.tsv(scaledTPM)
- multiqc_report.html : including fastQC reports and mapping rate of salmon(mapping rate for transcripts)
output sample
| Treg_LN_1 | Treg_LN_2 | |
|---|---|---|
| 0610005C13Rik | 0 | 0 |
| 0610006L08Rik | 0 | 1 |
| 0610009B22Rik | 4 | 10 |
| ... |
- output is scaledTPM (see. Soneson, C., Love, M. I. & Robinson, M. D. Differential analyses for RNA-seq: transcript-level estimates improve gene-level inferences. F1000Research 4, 1521 (2015).)。
—-gcBiasoption was added on salmon. You can refer to Mike Love's blog : RNA-seq fragment sequence bias.--validateMappingsflag was also adopted. (You can’t use it while using alignment-base mode.) Please see salmon Frequently Asked Questions for further details.- The reference for human is GENCODE Release 37 (GRCh38), and that for mouse is GENCODE Release M26 (GRCm38)
A serious bug was reported in the tximport_R.R and fixed. In the older version, Salmon's output and multiqc reports were correct and sometimes output.tsv were disturbed. Please update Ikra to the latest version. If you are using the old version(<1.1.1), please update and re-run ikra. We apologize for the inconvenience.
A bug has been reported that stops processing due to the following error in sra-tools.
docker: Error response from daemon: OCI runtime create failed: container_linux.go:345: starting container process caused "exec: \"fasterq-dump\": executable file not found in $PATH": unknown.
The latest version has already been corrected, so if you encounter the same error, please update to the latest version.
All you need is git clone ikra, and install docker or udocker(v1.1.3). No need for installing plenty of softwares! If you don’t want to use docker (or udocker), you must install all softwares by yourself and use —-without-docker option.
$ git clone https://github.com/yyoshiaki/ikra.gitif you use SRR mode, install sra-toolkit locally.
$ git pull origin master $ bash ikra.sh --version
...
ikra v2.0.1 -RNAseq pipeline centered on Salmon-
...- sra-tools : 2.10.9
- FastQC : 0.11.9
- MultiQC : 0.10.1
- Trim Galore! : 0.6.7
- Salmon : 1.4.0
- tximport : 1.6.0
- STAR : 2.7.8a
- Hisat2 : 2.2.1
- sambamba : 0.8.0
- deeptools : 3.5.1
- mouse:mm10 (GRCm38)
- human:hg38 (GRCh38)
SRR mode
$ cd test/Illumina_SE && bash ../../ikra.sh Illumina_SE_SRR.csv mouse --test -t 10fastq mode
You can execute it after you execute SRR mode. (That is because you don’t have fastq files.)
$ cd test/Illumina_SE && bash ../../ikra.sh Illumina_SE_fastq.csv mouse --fastq -t 10SRR mode
$ cd test/Illumina_PE && bash ../../ikra.sh Illumina_PE_SRR.csv mouse --test -t 10fastq mode
You can execute it after you execute SRR mode. (That is because you don’t have fastq files.)
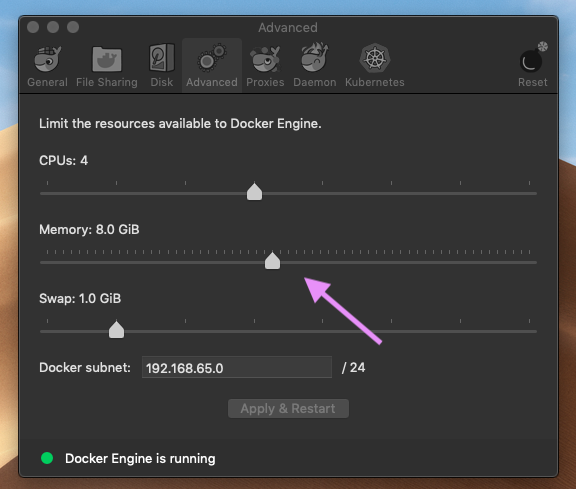
$ cd test/Illumina_PE && bash ../../ikra.sh Illumina_PE_fastq.csv mouse --fastq -t 10cd test && bash test.sh && bash test.full.shDr.Ota(DBCLS) solved the problem that salmon doesn’t work on Mac. The cause of the problem is that Docker is allocated only 2GB by default on Mac. The problem will be solved by allocating sufficient memory space(>=8Gb) for Docker, and applying and restarting Docker.
You can find SRR data so quickly in http://sra.dbcls.jp/
- In exporting output.tsv to iDEP, which data type should I select?
When iDEP reads output.tsv, please put a check to Read counts data.
Please refer to issue
Please refer to Relases
- add support for udocker
- add setting of species
- gtf and transcript file from GENCODE
- salmon
- trimmomatic(legacy)
- trim_galore!
- tximport
- fastxtools(for Ion)
- judging fastq or SRR(manual)
- introduce "salmon gcbias correction"
- salomn validateMappings
- pigz(multithread version of gzip)
- fasterq-dump
- cwl development is in progress
- rename to "ikra"
- protein coding option
Moved the flow using trimmomatic to ./legacy
2019/03/22 https://youtu.be/weJrq5QNt1M We tried developing it because Mr.Michael visited Japan. For now, cwlnized trim_galore and salmon in PE.
cd test/cwl_PE && bash test.sh
Hiraoka Yu, Yamada Kohki, Ryuichiro Yamsasaki, YusukeKawasaki, Kitabatake Ryoko, Matsumoto Yasunari, Ishikawa Kaito, Umezu Yuto, Hirose Haruka, & Yoshiaki Yasumizu. (2021). yyoshiaki/ikra: ikra v2.0.1 (v2.0.1). Zenodo. https://doi.org/10.5281/zenodo.5541399
This software is freely available for academic users. Usage for commercial purposes is not allowed. Please refer to the LICENCE page. If you are not an academic user, please contact to the author.